Isotopic measurements of nitrogen, chlorine and other elements help trace pollutants in streams and groundwater. For example, precise measurements of the abundances of carbon isotopes are used to determine purity and source of food, such as honey and vanilla. These small variations in an element's atomic weight can weigh heavily on research and industry. For example, sulfur is commonly known to have a standard atomic weight of 32.065, but its real atomic weight can be anywhere between 32.059 and 32.076, depending on where the element is found. However, the abundance of an isotope can vary in nature, leading to variations in an element's atomic weight. (The numbers in each isotope reveal how many particles they have in their nuclei - carbon-12 has six protons and six neutrons.) In the past, to give a standard atomic weight for these elements, scientists averaged out the atomic weights of these isotopes based on how common those isotopes are - the more plentiful an isotope was, the bigger a role it played in the standard atomic weight. 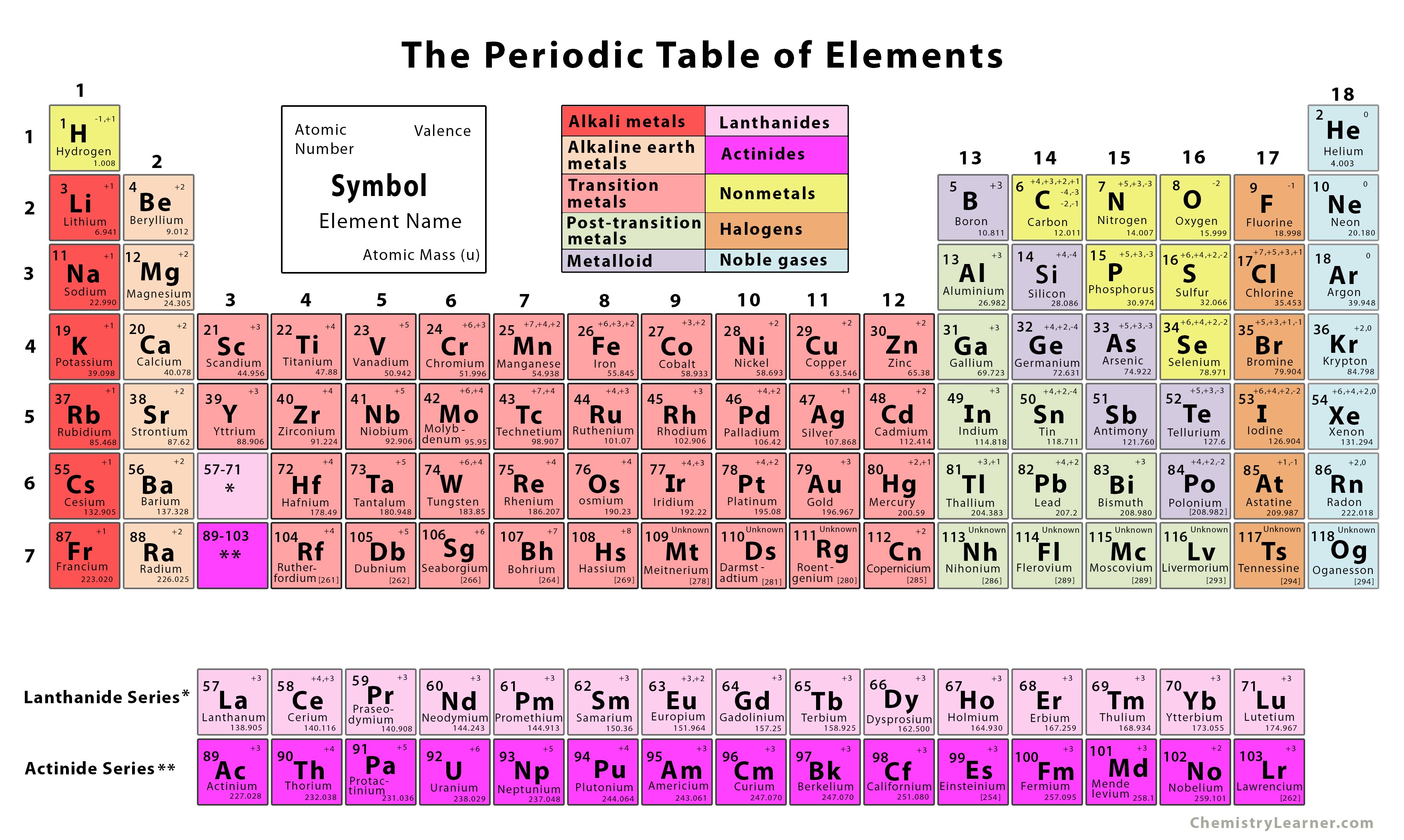
For instance, carbon has two - carbon-12 and carbon-13. /chemical-periodic-table-of-elements-with-color-cells-vector-illustration-840464844-ff61af51081b4fb1959504105e232e01.jpg)
While all the atoms making up an element have the same number of protons, elements have variants known as isotopes that possess different numbers of neutrons in their nuclei, making some lighter or heavier than others.Ĭertain elements have more than one stable isotope. The problem that scientists are now addressing is rooted in the fact that these atoms don't always have the same masses. The standard atomic weight of an element, which is made up of one type of atom, is based on the mass of its atoms. "As technology improved, we have discovered that the numbers on our chart are not as static as we have previously believed." "For more than a century-and–a-half, many were taught to use standard atomic weights - a single value - found on the inside cover of chemistry textbooks and on the periodic table of the elements," said physicist Michael Wieser at the University of Calgary. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
